The Challenge
Each market required a different portal structure based on its country regulations. Content such as educational materials, therapy areas and product information could be visible, restricted or require authentication depending on the region.
Designs were previously managed in separate country-specific files, with variations across each section. This made it difficult to update and maintain, and increased the risk of compliance issues - a costly process if the designs did not meet regulatory requirements.
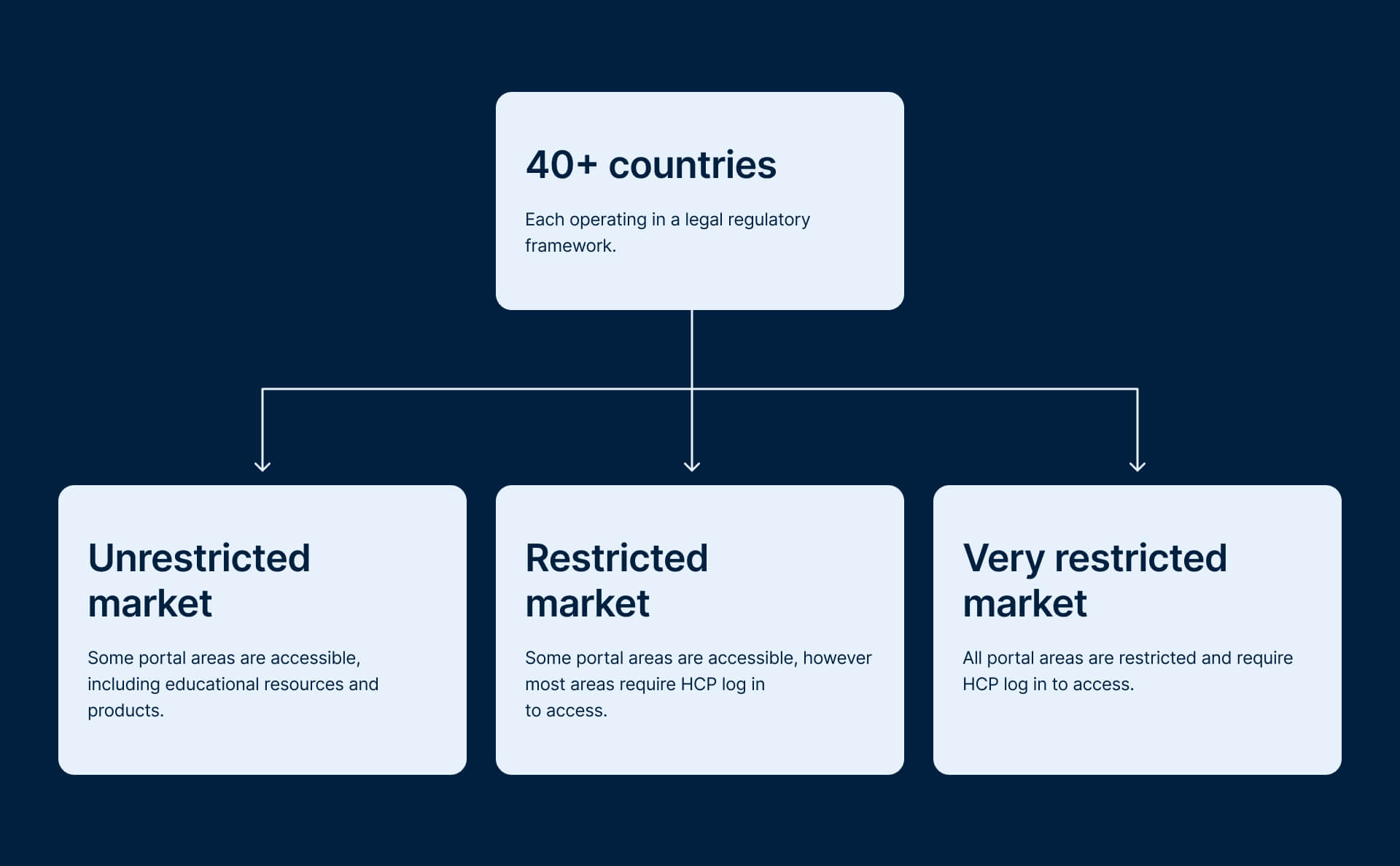
Framework Approach
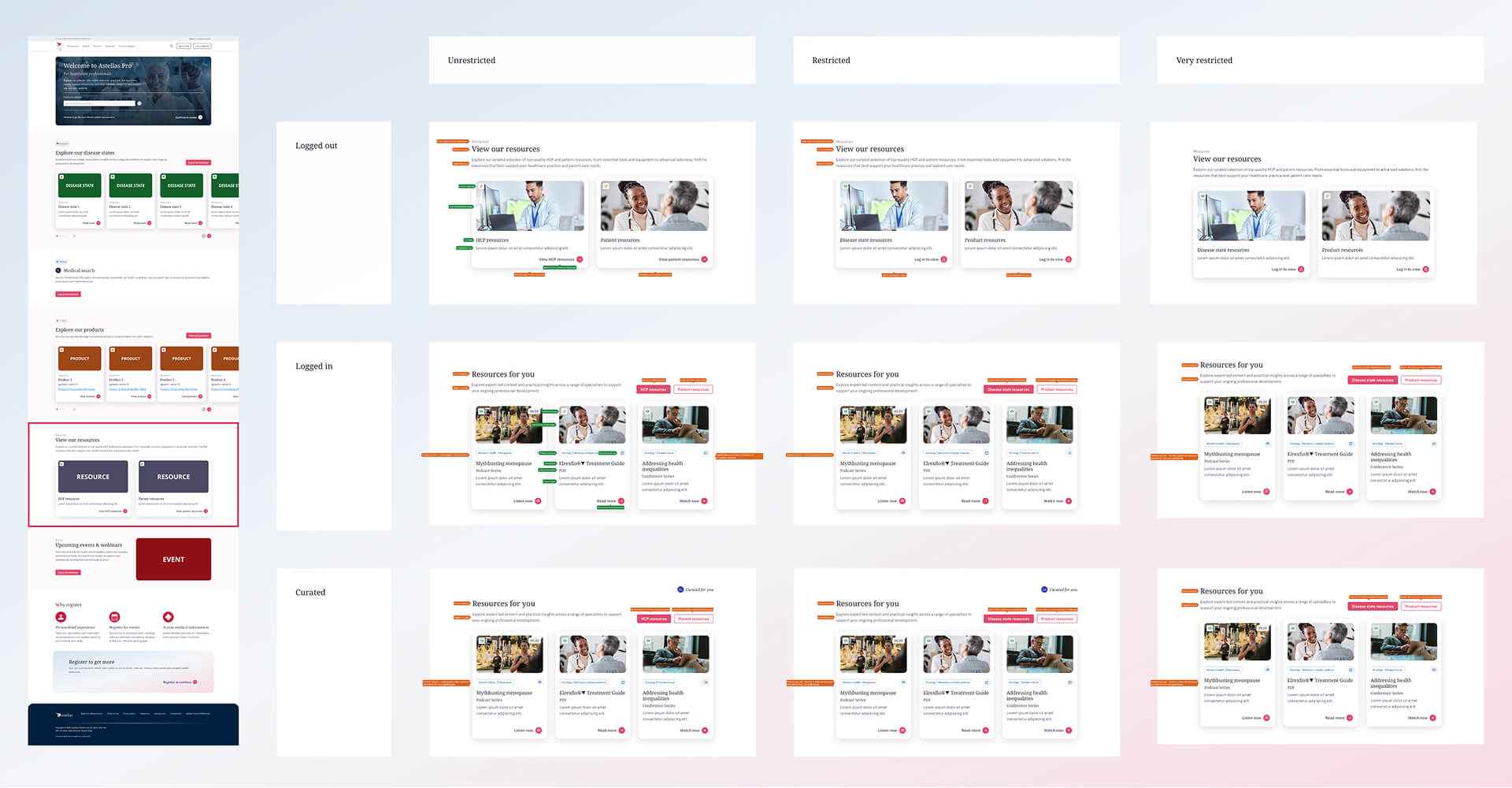
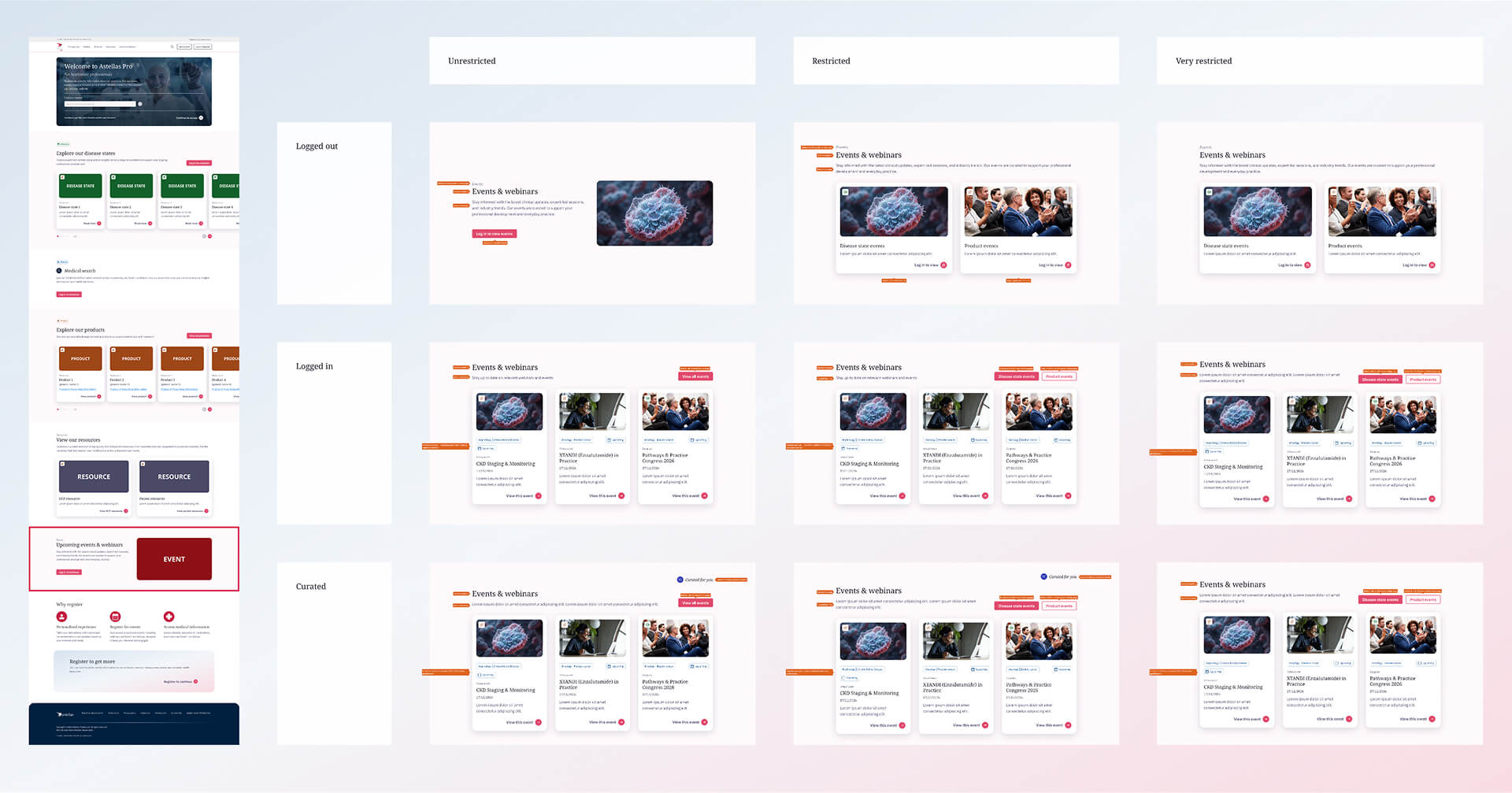
To simplify this, I was tasked to restructure the system around three market types: unrestricted, restricted, and very restricted.
This shifted the approach from designing per country to designing based on rules, making it easier to understand what content could be shown in each section.
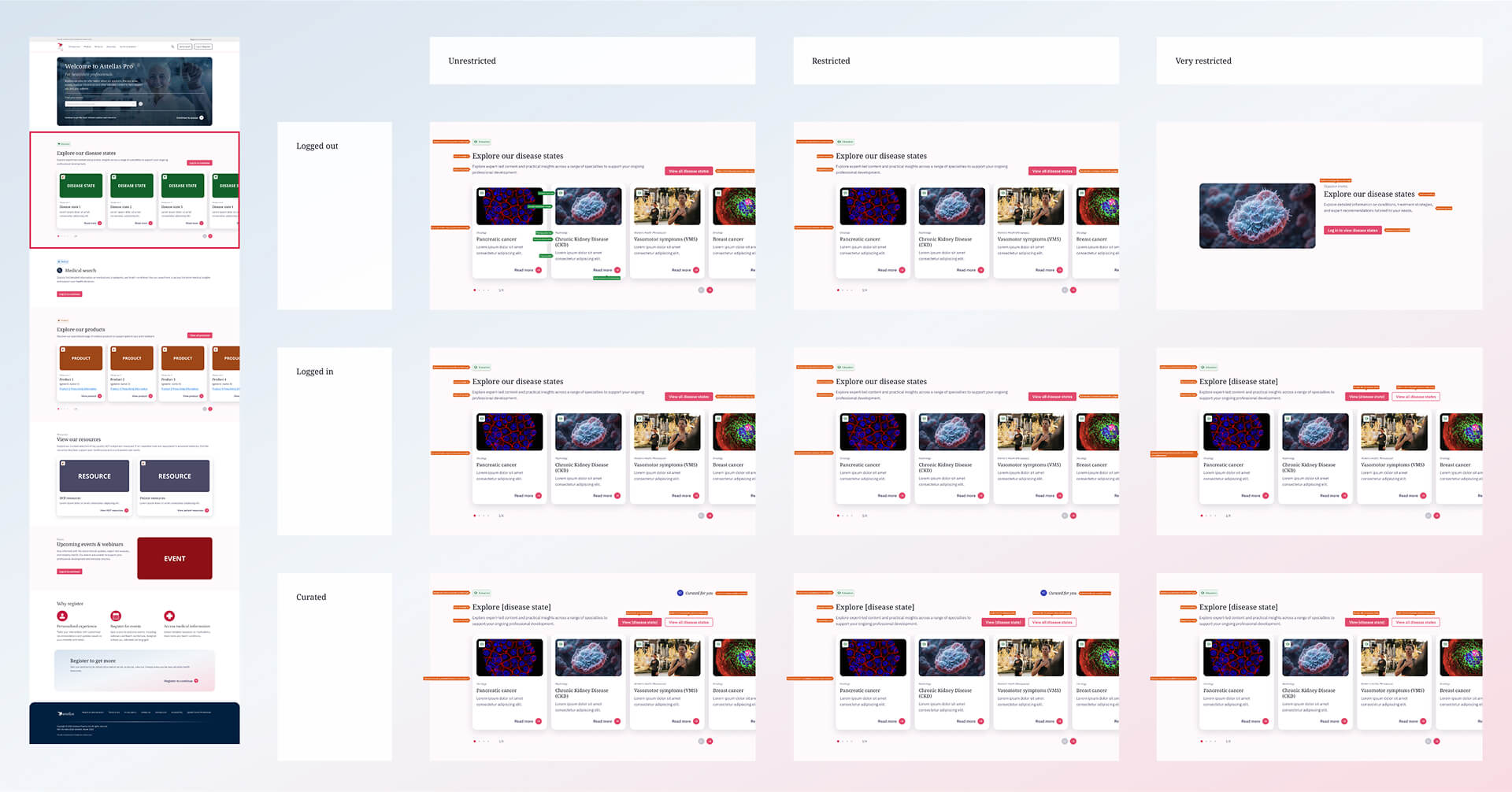
Disease state section
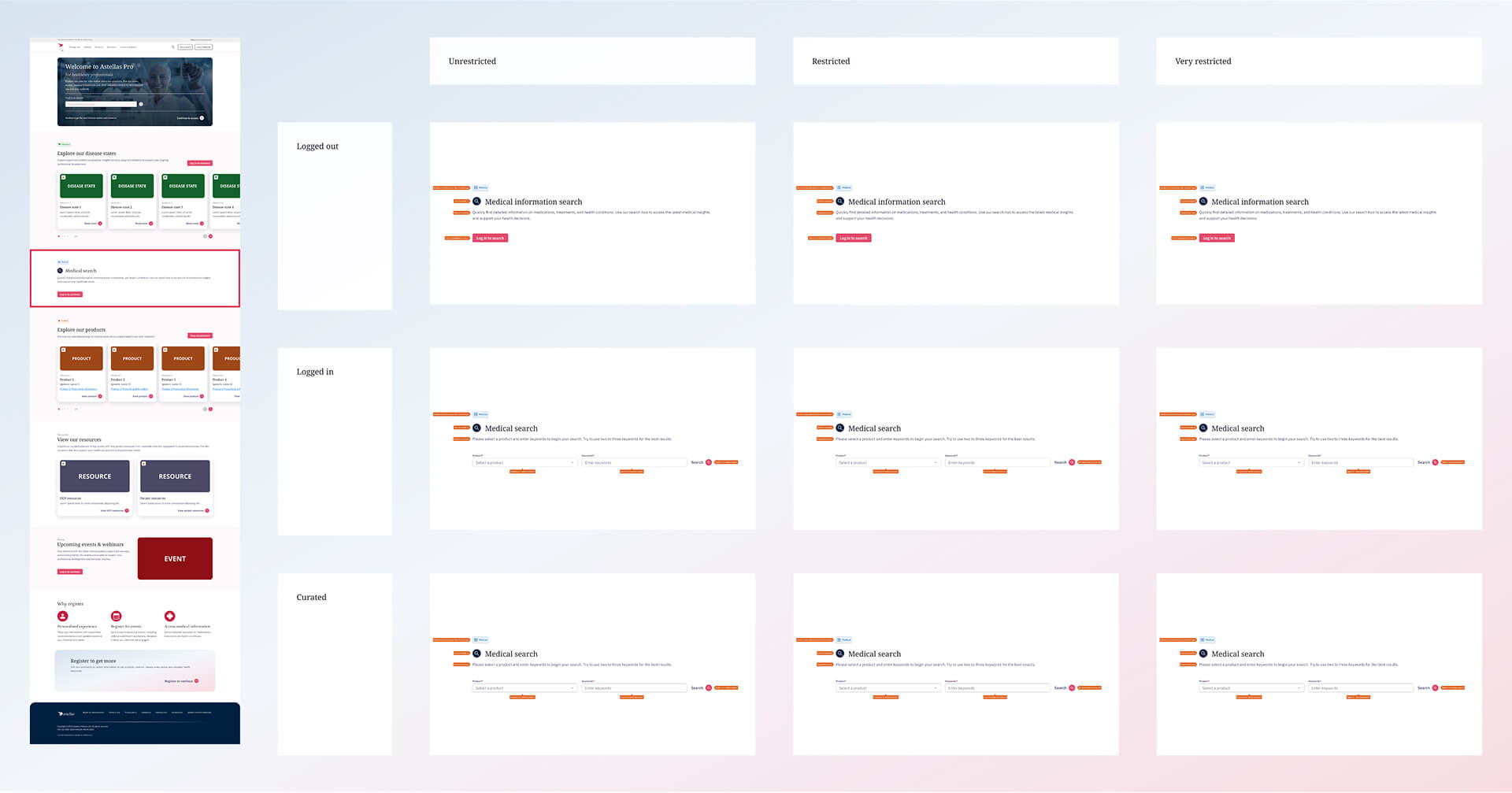
Medical information section
Products section
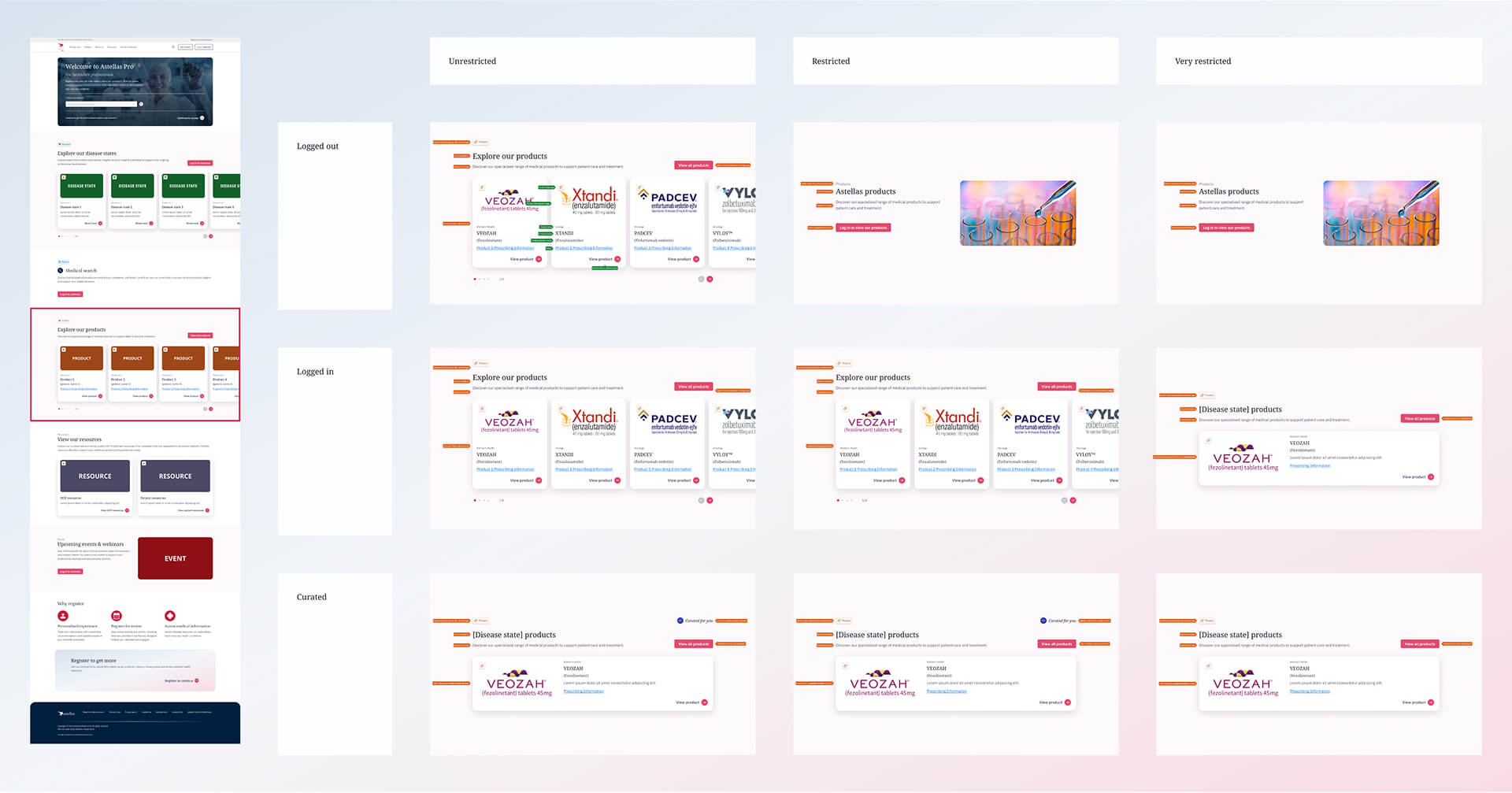
Resources section
Events & Webinars section
The outcome
The framework was built into a modular Figma library, with components and templates aligned to each market type.
Designers could assemble landing pages using predefined section variations, rather than maintaining multiple files per country. This created a single source of truth and improved consistency across markets.
By grounding designs in pre-defined rules, teams could work more efficiently and with greater confidence that each section and its linked experience met regulatory requirements. This reduced review time and minimised costly revisions with legal and medical teams.